Validation Services
TURN-KEY, COST-EFFECTIVE TEMPERATURE AND HUMIDITY MAPPING VALIDATION SOLUTIONS
Parameter has extensive experience in the development and execution of industry-proven stability chamber validation protocols. Parameter is equipped to provide and execute stability chamber validation protocols such as Installation, Operational and Performance Qualification.
We collect all temperature data with the highest quality equipment, including a Rotronic HW4 Mapping System and digital Rotronic HC2 dual Temperature and Humidity sensors, allowing for temperature and humidity measurements at each mapped point within the chamber. All test equipment calibration is NIST traceable.
TEMPERATURE MAPPING VALIDATION DOCUMENTATION
Parameter provides a complete document package tailored to your needs with each temperature mapping validation. We can produce a complete mapping protocol for you, or follow a protocol that you have already created. If you don’t need a full protocol for your temperature and humidity mapping study, we can provide a basic stability chamber validation plan prior to performing the study to specify the mapping details.
Regardless of the level of documentation you choose, after the mapping study has been completed you will be provided a clear comprehensive summary of the results.
Document Package results include:
- Minimum, Maximum and Average Statistics
- Mean Kinetic Temperature calculations
- Detailed graphs
- Test data
- Deviations
- The executed protocol
- Test equipment calibration data with NIST-traceable calibration certificates.
Chamber Mapping studies include:
- IQ/OQ/PQ
- Empty Chamber Mapping
- Full / Loaded Chamber Mapping
- Simulated Power Outage
- Open Door Recovery
- Alarm Testing
- Single and Multi-point Conditions
- Calibration Verification
Once execution is complete, a stability chamber validation report package is carefully prepared, reviewed and submitted for client approval.
Our Validation Engineers have extensive experience with temperature and humidity stability chambers and can provide you with the high quality service that will assure sample integrity and satisfy the most stringent regulatory requirements. Our ultimate goal is to leave you with the peace of mind that your company’s validation requirements are being met.
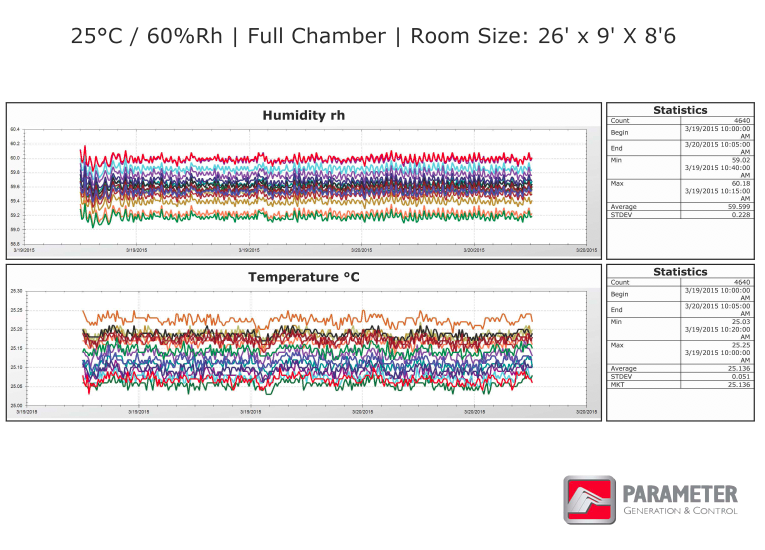
STABILITY CHAMBER QUALIFICATION GRAPHS
These graphs show validation data performed on a large walk-in room, once while empty and again while full. Each color represents a different sensor located throughout the room. Notice the standard deviation rates for the entire data set represented on the right of the graphs, indicating that the walk-in room is being controlled very tightly throughout the validation cycles.